When prevention fails: prescription is inevitable
Listen to the article now
Audio by The Catalyst
00:00 / 00:00
Series: The Fragile Powertrain of Antimicrobial Stewardship: Infection Prevention and Control as the Powertrain’s Chassis
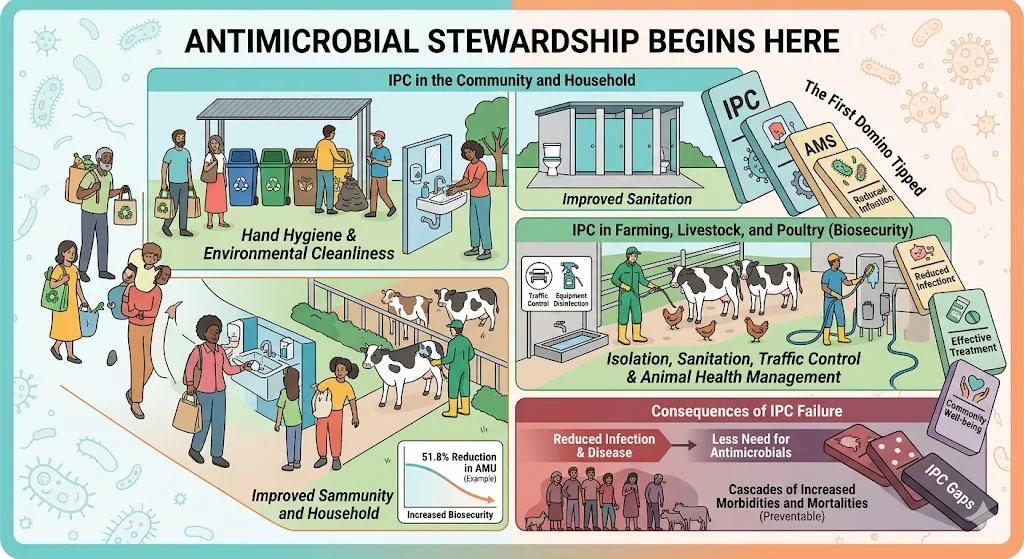
The first domino in Antimicrobial Stewardship (AMS) has already tipped and initiated a cascade of events by the time the patient enters a hospital hallway seeking medical treatment. AMS does not begin in the consultation room when the doctor writes the prescription, despite the common misconception, it starts outside the hospital walls: in our homes, farms, and our environment through effective infection prevention and control (IPC) practices in each of these settings. Effective infection prevention and control practices encompass precautions to prevent infection, interrupt transmission, promote education, and ensure continuous monitoring and improvement of practices (1). Due to the foundational role IPC plays in Antimicrobial Stewardship, understanding how IPC practices operate at the household and community level is crucial to achieving better antimicrobial stewardship.
IPC in everyday settings: Households and community 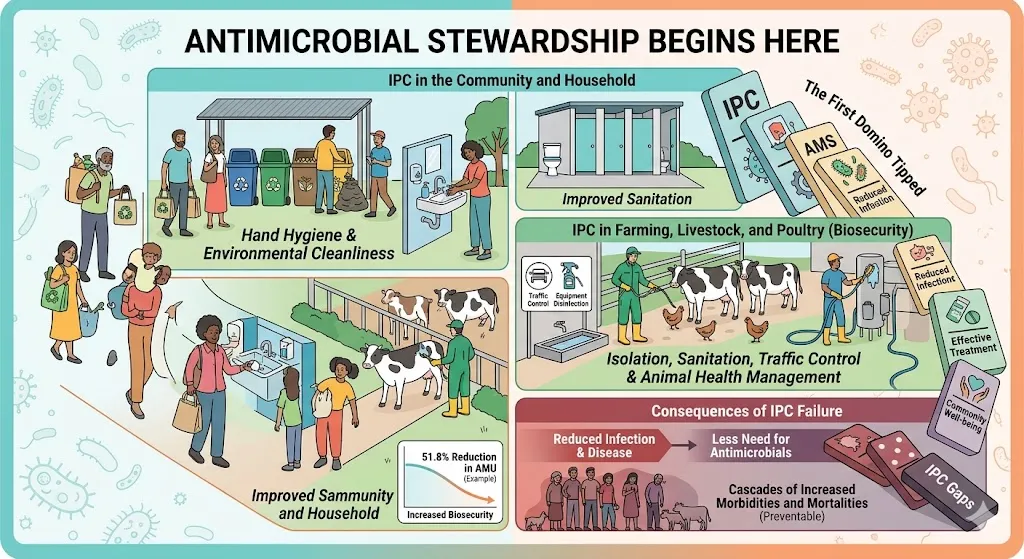
In our day-to-day home lives, we are consistently engaged in infection prevention and control. The implementation of these practices varies depending on the infrastructure available, individual knowledge, institutional policies/guidelines, cultural and social context, and others. At the Community level, infection prevention and control practices typically comprise basic public health and hygiene practices such as hand hygiene, proper waste disposal, and environmental cleanliness.
For hand hygiene, it is mainly practiced in the following circumstances: after toilet use, after changing diapers, before food handling, before eating, and after coughing or sneezing. These practices have minimal requirements that are: reliable water supply, soap or alcohol based hand rub (ABHR), clear information on why, when, and how to clean hands, and a conducive physical and social environment. However, the full implementation of these basic hygiene practices is still quite limited worldwide [2]. This could be attributable to infrastructural and socio-economic resource constraints [3,4]. In 2024, it was estimated that 1.7 billion people lacked basic hand hygiene services at home, and 611 million had no handwashing facilities at all (2). This means that while one can have the will and the knowledge to practice hand hygiene, they may be unable to, and this creates an environment where infections easily spread.
On the other hand, in settings where supplies are available, challenges still arise, namely, inconsistent technique and lack of motivation to apply approved hand hygiene practices. In most cases, these instances happen, especially after using the washroom or before eating, where one can underestimate the need for thoroughness in handwashing; this can be due to perceived cleanliness of their hands or because one uses utensils to eat food, and not hands. In such scenarios, handwashing is often performed like a sprint, fast, almost frantic, without thoroughness. While this may seem efficient, it undercuts the very purpose of proper hand hygiene, which requires thorough hand washing for effectiveness in disease prevention. This practice, without thoroughness, transforms from being protective to being symbolic.
Beyond our personal hygiene, environmental conditions also pose undeniable risks depending on the level of cleanliness. These conditions are often evident in places where garbage is strewn across fields, gradually accumulating into small hills that become garbage mounds, or where roadside trash piles into the water drainage trenches, slowly but surely blocking the water drainage trench and creating stagnant water. This situation could be the result of a lack of waste collection services, public dustbins, or bins in private and public vehicles. This is a significant concern, as approximately 2 billion people worldwide live without waste collection services, and waste generation in lower-income cities in Africa is projected to double by 2030 ( 5).
Adding to these risks, over 1.5 billion people worldwide lack access to private toilets or latrines, and 419 million continue to practice open defecation into street gutters, behind bushes, or into open bodies of water (6). In Kenya, according to the 2019 census, it was revealed that 7.4% of households lacked proper sanitation facilities and approximately 10% of the population practiced open defecation (7). In such an environment with trash piling into mounds, blocking water trenches, and open defecation, we collectively create a reservoir for pathogens to live in and multiply, thereby serving as a potential source of infection.
These statistics are not peripheral: they expose structural vulnerabilities and a normalized lack of care for environmental cleanliness that extends beyond our compounds. This creates an environment that is conducive to the thriving of infectious agents, promotes disease transmission, and consequently drives an increase in antimicrobial consumption. Given the critical impact of environmental cleanliness on public health, it is equally important to address infection prevention and control (IPC) in farming, where close human-animal interactions and poor biosecurity measures pose additional risks for disease transmission.
IPC in farming, livestock, and poultry.
With these risks in mind, it is important to consider Infection Prevention and Control practices in farming. Farming may not involve everyone directly, but we all benefit from or are affected by the current farming practices in operation. Thus, we can’t overlook IPC in farming, which is commonly referred to as biosecurity measures. Biosecurity includes all measures to prevent the introduction of pathogens (bio-exclusion) and reduce the spread of pathogens (bio-containment) (8). While the principles remain similar to human Infection Prevention and Control practices, the methods differ slightly. In biosecurity, the focus is on four key elements to maintain livestock health: isolation, traffic control, sanitation, and animal health management (9)
These biosecurity elements are interconnected and work synergistically. Within the farming premises, isolation serves as the first line of defense, aiming to prevent the introduction of new pathogens. This is achieved by separating new or returning animals from the existing stock, as well as separating sick animals from healthy ones.
Building on the first measure, we have traffic control, which is practiced within the farm premises to reduce the creation of any opportunities for new pathogens to be introduced and spread. This is accomplished through measures such as fencing the farm and having one single point of entry into the farm, disinfecting equipment and vehicles before entry, limiting the number of people accessing the farm, and requiring those who do enter to wear dedicated clothing when interacting with the animals. Additionally, traffic control extends to animal movement within the premises through the application of the all-in, all-out principle, which allows for the complete disinfection of farm premises before introducing a new batch of animals, ensuring the environment does not serve as a potential reservoir for pathogens.
While the measures above focus on preventing the introduction and spread of new pathogens, sanitation compounds their effectiveness through maintaining a clean premise and thus eliminating potential reservoirs where pathogens could persist and undermine the effectiveness of the other measures.
The last measure, animal health management, focuses on enhancing the animals’ innate immunity and reducing factors that predispose them to morbidities. This is achieved through a combination of preventive and curative strategies such as proper housing, high-quality feed and water, vaccination, and routine health monitoring to detect and manage illnesses early. Together, these measures significantly reduce disease incidence and therefore minimize the reliance on antimicrobials within the farming industry. Research supports this connection , for instance, a review of 27 peer-reviewed studies across European countries demonstrated a 51.8% reduction in antimicrobial use linked to enhanced farm biosecurity practices(10). Although some studies do not quantify the exact decrease, they consistently report a lowered need for antibiotic use due to reduced morbidity as a result of effective implementation of biosecurity measures (11,12,13,14,15)
However, the benefits of biosecurity measures can only be fully actualized through proper implementation. Poor or inconsistent application undermines their effectiveness, increasing morbidity rates and increasing reliance on antimicrobials. This is a pattern commonly observed in several studies. Examples from African countries such as Ethiopia, Kenya, Cameroon, and Uganda highlight this tendency to substitute antimicrobials for effective biosecurity practices.
In Ethiopia, studies on dairy cow biosecurity revealed poor implementation, particularly in the following measures: sanitation and animal health management (9). Similarly, in Cameroon, sanitation efforts, especially vector control measures and measures ensuring a water and feed quality, were inadequate (16). In Uganda, while health management practices like vaccination, stocking density, and management of sick birds had relatively high compliance at 61%, other biosecurity measures performed poorly by a huge percentage. For instance, cleaning and disinfection of farm premises was only at 33.1%, and traffic control measures, including fencing and visitor protocols, were even lower at 20.8% (17).
Kenya presents a mixed picture based on two separate studies. One, covering three counties: Kajiado, Kiambu, and Machakos counties, found overall biosecurity rated at 67.9%. Measures preventing pathogen introduction, such as visitor and worker protocols, purchase of day-old chicks, and feed and water management, were relatively strong at 42.3%. However, practices aimed at limiting pathogen spread within farms, like sanitation and disease management, were low at 25.6% (18). Another study in Nyanza reported poor implementation across all measures but animal health management. Adoption rates for practices like deworming ranged from 36.2% to 78.7%, vaccination from 67.6% to 97.9%, and carcass disposal from 86% to 91.5%. Conversely, sanitation measures ranged from 13.5% to 45.9%, with routine sanitization as low as 0.3% to 25.6%, though cleaning equipment with soap fared better (53.5% to 76.5%). Traffic control practices such as foot disinfection and use of dedicated clothing were poorly adopted, ranging from 2.2% to 37.3% and 1.6% to 30.3%, respectively(19).
These findings could suggest that farmers often rely on antimicrobials as a crutch or back-up for inadequate biosecurity, using antibiotics prophylactically and giving uneven priority to different biosecurity measures. Regional variations in prioritization may reflect cultural differences or infrastructure challenges. Overall, biosecurity does not appear to be a central focus in farm management, with adoption influenced by perceived cost, risk, and importance. This raises important questions: Are cultural practices influencing which biosecurity measures are prioritized and which ones are overlooked, or could it be that financial constraints are the main factor driving these trends?
Reflecting on these patterns prompts critical inquiry: How much of the modern dependence on antimicrobials stems not from microbial evolution, but from our collective failure to implement effective biosecurity? And how do these gaps perpetuate cycles of infection that affect communities, economies, and health systems?
Barriers to effective Infection Prevention and Control implementation
At the community level, effective Infection Prevention and Control (IPC) is hindered by gaps in knowledge, lack of adequate infrastructure, and social norms that shape how risks and benefits are perceived. Where infrastructure exists and IPC behaviors are culturally supported, compliance is generally higher. Conversely, inadequate facilities combined with socially accepted unhygienic practices, such as open defecation, often reduce the practice of efficient IPC to inconsistent, individual choices (20).
In farming, reported barriers to implementation of biosecurity measures included: high cost, increased workload, perception of limited usefulness of biosecurity measures, and unclear and insufficient knowledge. (10,19). Interestingly, having a larger number of livestock was not always seen as a barrier, possibly because higher livestock numbers are often perceived as a commercial venture with higher economic risks (19), motivating the farmer to adopt effective biosecurity measures. In contrast, small-scale farmers may delay investing in biosecurity measures until losses occur, at which point implementation becomes reactive rather than preventive.
This reactive pattern extends beyond farming into both household and community settings, where infection prevention and control measures seem to be enacted in response to problems after they arise. In environments lacking support, infection prevention shifts from a collective responsibility to a personal burden. This shift raises public health questions about bridging the chasm between individual agency and systemic support to make prevention not a privilege but a foundational societal norm.
The bridging in farming could perhaps start with shifting from treating biosecurity as an afterthought to viewing it as an investment in resilience against inevitable ecological disruption.
Consequences of Infection Prevention and Control Failure
When Infection prevention and control practices aren’t effectively implemented, they fail, the domino tips, and the resulting domino effect are consequences, which affect not just the farmers but the entire community. For the community, poor Infection Prevention and Control practices mean initiating a cascade of increased morbidities, increased mortalities, especially in vulnerable populations, higher hospitalization rates, and all these then lead to greater economic burdens in households due to increasing admission rate in hospitals, which increases unaccounted household expenditure and effectively removes someone from the workforce. Even in scenarios where one isn’t admitted, receiving medical treatment is costly through the diagnostic tests needed and medication used to relieve and or cure ailments.
For example, diarrheal diseases remain the third leading cause of death among children aged 1 to 59 months globally despite being largely preventable and treatable. Each year, approximately 443,832 children under 5 and 50,581 children aged 5 to 9 years die from diarrheal diseases(21). The gap can be solely attributed to biomedical limitations, considering advances such as rotavirus vaccination and affordable and available treatments like oral rehydration salts and zinc supplementation, but also stems from inadequate preventive infrastructure. Lack of safe drinking water, inadequate sanitation, and poor hygiene are primary factors contributing to these preventable deaths. These preventable losses compel us to question how many futures are forfeited not as a result of these pathogens alone, but the inertia of an inadequate system that has failed to prioritize the foundational safeguards to health.
For farmers, poor biosecurity causes reduced livestock productivity, affects the quality of the animal products, increases livestock morbidities, mortalities, and avoidable treatment costs. Poor farm biosecurity also threatens community health. Many poultry diseases are transboundary, which means diseases that are highly contagious with rapid cross-border spread and significant socio-economic and public health consequences (22). Thus, poor biosecurity shifts from not being just an economic risk to the affected farm level, but a regional and global threat. Addressing barriers will require systemic thinking: How do we align economic realities with preventive imperatives to avert cascades that threaten food security and public health?
Recommendations and success stories
In public health, we have a recent and gradual shift from the traditional, siloed antimicrobial stewardship approach to a one-health framework. This approach presents an opportunity to apply Infection Prevention and Control (IPC) as a cross-cutting strategy across all sectors and moving away from the reactive, hospital-centered model toward a proactive, community-based system. Small changes at the community level can produce sustainable long-term benefits. Examples of such practices include culture shifts like washing hands in a common basin to washing with running water for at least 20 seconds. Such a small change improves hand hygiene effectiveness. When it comes to respiratory hygiene, a shift to coughing or sneezing into a disposable tissue or into the elbow instead of what is commonly done, which is coughing into one's palm or covering your face with the palms when sneezing, reduces pathogen transmission through reducing hand contamination and containing possible infectious pathogens expelled by coughing or sneezing. For the prevention of reservoir formation in our environment, regularly cleaning and disinfecting frequently touched surfaces such as toilets, bathrooms, and kitchens is effective.
In addressing infrastructural challenges affecting hand hygiene, simple innovations also play a crucial role, as was seen during the COVID-19 pandemic. One such innovation is the Tippy Tap, which provides a reliable water supply necessary for proper hand hygiene in resource-limited settings by using minimal materials (23).
In farming, some measures that can improve the effectiveness of a farm’s biosecurity are: quarantining sick animals and assessing the health status of animals before purchase, separating young and newborn animals to avoid cross-contamination, cleaning and disinfecting farm equipment and vehicles, using dedicated old clothes and boots in good condition, practicing an all-in-all-out principle for the animals, and fencing properties to separate livestock from wild or other animals.
Other additional and simple solutions involve using foot baths at coop entrances, cleaning and disinfecting water troughs, feeders, and premises, prompt separation of the animal carcasses from the living animals, and sanitizing equipment and areas used for carcass disposal after contact. Wherever it is possible, having separate equipment and designated areas for carcass disposal and disinfection after each use is recommended.
In improving an animal’s innate immunity and maintaining its health, it is important to provide well-ventilated housing, reduce stock density, vaccinate and deworm according to schedule, increase weaning age, and routinely monitor for illness.
Several documented success stories have demonstrated the feasibility and benefits of focusing on prevention through IPC practices. Starting with Kibera in Nairobi county, where the introduction of waterless hand sanitizer in schools increased hand hygiene compliance after latrine use and before meals to 82%, leading to reductions in diarrheal incidence and school absenteeism (24). In Kilifi County, there was the Community-Led Total Sanitation (CLTS) initiative, which reduced open defecation by involving the community, transforming IPC from an individual burden to a shared responsibility (20). In Kisumu (2018–2021), a project funded by Practical Action and the UK government extended water pipelines by 18.5 km, improved water access for 3,966 households, and benefited 19,830 people by addressing inadequate infrastructure for proper hand hygiene (25). Meanwhile, in Tanzania, the “Mikono Safi” school-based hand hygiene education program improved knowledge, skills, and normative reinforcement, effectively addressing the “why,” “how,” and “when” for proper hand hygiene (26).
While sustained small changes can lead to significant improvements, governance support is also critical. In Kenya, key strides in IPC began with establishing IPC guidelines and County Infection Prevention and Control Advisory Committees (CIPAC) in 23 counties. Additionally, 40 veterinary officers received biosecurity training, and 318 farmers across Nyeri, Machakos, and Nakuru were trained in broiler production, antimicrobial use, and biosecurity (27).
These examples demonstrate that in a community where infrastructure, education, and social reinforcement align, IPC behaviors can shift measurably. They also provoke reflection on what scalable insights from localized successes could inform a global paradigm where prevention of illnesses is embedded into the fabric of daily life and production, instead of our reactive approach of managing illnesses. Yet achieving this paradigm requires first dismantling the prevailing notion that disease prevention and antimicrobial stewardship are concerns that belong and begin only within clinical settings.
Conclusion
There remains a persistent misconception about where exactly antimicrobial stewardship begins. This misconception is that the stewardship begins in the hospital hallways, but the reality is that by the time a prescription is written, multiple preventive opportunities have already been missed in our farms, homes, and environment. If antimicrobial stewardship is a powertrain guiding us to a future where antimicrobials remain effective, Infection Prevention and Control is the chassis providing structural integrity of this powertrain. When it is weak, downstream interventions become compensatory rather than preventive, and the costs, whether it's human lives or economic burden, are borne by the affected communities and society at large.
Strengthening Infection Prevention and Control will require coordinated investment and a one- health approach in infrastructure, behavioral reinforcement, agricultural biosecurity, and community-level systems. Prevention must become the default rather than reactive. There is potential for future research to focus on context-specific implementation strategies that strengthen this structural foundation and shift stewardship from reactive containment to upstream prevention. As we contemplate the intersections of human, animal, and environmental health, the imperative is clear: fortifying IPC is not merely a technical endeavor but a scientific commitment to sustainable well-being in our communities.
- World Health Organization. Guidelines on core components of infection prevention and control programmes at the national and acute health care facility level. Geneva: World Health Organization; 2016.
- UNICEF, World Health Organization. Progress on household drinking water, sanitation, and hygiene 2000–2024: special focus on inequalities. New York: UNICEF; 2025.
- Muramatsu-Noguchi Y, Nonaka D, Kounnavong S, Kobayashi J. Association Between Socio-Economic Status and the Presence of Soap at Handwashing Facilities in Lao People's Democratic Republic: A Cross-Sectional Study. Asia Pac J Public Health. 2022 May;34(4):423-426. doi: 10.1177/10105395211072478. Epub 2022 Jan 18. PMID: 35040349; PMCID: PMC9131397. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9131397/
- Simiyu SN, Busienei PJ, Mbaya N, Baker KK, Dreibelbis R, Cumming O. Determinants of access to basic handwashing facilities and handwashing with soap in low-income areas of four Kenyan cities. PLOS Glob Public Health. 2025 Jul 17;5(7):e0004921. doi: 10.1371/journal.pgph.0004921. PMID: 40674324; PMCID: PMC12270180. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC12270180/
- World Bank. What a waste 2.0: a global snapshot of solid waste management to 2050. Washington (DC): World Bank; 2018.
- World Health Organization, UNICEF. Progress on household drinking water, sanitation, and hygiene 2000–2022: special focus on gender. Geneva: WHO; 2023.
- Kenya National Bureau of Statistics. 2019 Kenya population and housing census volume IV: distribution of population by socio-economic characteristics. Nairobi: KNBS; 2019.
- Renault V, Humblet MF, Saegerman C. Biosecurity concept: origins, evolution and perspectives. Animals (Basel). 2021. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC8749630
- Harun MH, Shimelis S, Andargie B, Abdi MY, Bekere HY. Assessment of biosecurity status in dairy cow farms. Bull Natl Res Cent. 2022;46:152. doi:10.1186/s42269-022-00836-7.
- Laanen M, Persoons D, Ribbens S, et al. Relationship between biosecurity and production/antimicrobial treatment characteristics in pig herds. Vet J. 2013;198(2):508-512. doi:10.1016/j.tvjl.2013.08.029.
- Moiane B. The Role of Biosecurity in Promoting Farm Animal Welfare in Low- and Middle-Income Countries [Internet]. Veterinary Medicine and Science. IntechOpen; 2025.Available from: https://doi.org/10.5772/intechopen.114891
- Postma M, Backhans A, Collineau L, Loesken S, Sjölund M, Belloc C, Emanuelson U, Grosse Beilage E, Nielsen EO, Stärk KDC, Dewulf J. Evaluation of the relationship between the biosecurity status, production parameters, herd characteristics and antimicrobial usage in farrow-to-finish pig production in four EU countries. Porcine Health Management. 2016;2:9. doi:10.1186/s40813-016-0028-z. Available from: https://www.researchgate.net/publication/297752459_Evaluation_of_the_relationship_between_the_biosecurity_status_production_parameters_herd_characteristics_and_antimicrobial_usage_in_farrow-to-finish_pig_production_in_four_EU_countries
- Descamps F, Dreesen L, Sunderland S. Vaccines and antimicrobial resistance: a veterinary pharmaceutical industry perspective. Philosophical Transactions of the Royal Society B: Biological Sciences. 2026;381(1944):20250103. doi:10.1098/rstb.2025.0103. Available from : https://www.researchgate.net/publication/400942612_Vaccines_and_antimicrobial_resistance-a_veterinary_pharmaceutical_industry_perspective
- Mallioris, Panagiotis & Dhaka, Pankaj & Makovska, Iryna & Sabben, N & Wagenaar, J & Stegeman, Arjan & Mughini-Gras, Lapo & Dewulf, Jeroen. (2025). Improved animal husbandry, biosecurity and vaccination as a strategy to reduce antimicrobial use. 10.20506/rst.44.3682.
- Pinto, Chris & Keestra, Sarai & Tandon, Pranav & Cumming, Oliver & Pickering, Amy & Moodley, Arshnee & Chandler, Clare. (2023). Biosecurity and water, sanitation, and hygiene (WASH) interventions in animal agricultural settings for reducing infection burden, antibiotic use, and antibiotic resistance: a One Health systematic review. The Lancet Planetary Health. 7. e418-e434. 10.1016/S2542-5196(23)00049-9. Available from: https://www.thelancet.com/journals/lanplh/article/PIIS2542-5196(23)00049-9/fulltext
- Ziebe SD, Vougat Ngom R, Akoussa AMM, Bogning HP, Zangue HA. Impact of Biosecurity on Production Performance and Antimicrobial Usage in Broiler Farms in Cameroon. Animals (Basel). 2025 Jun 16;15(12):1771. doi: 10.3390/ani15121771. PMID: 40564323; PMCID: PMC12189605. Available from: pmc.ncbi.nlm.nih.gov/articles/PMC12189605/#sec3-animals-15-01771
- Ayebare, D., Mbatidde, I., Kemunto, N.P., Muloi, D.M., Ibayi, E.L., Nielsen, S.S., Ndoboli, D., Roesel, K., Tenhagen, B.-A. and Moodley, A. 2025. Biosecurity measures and effects on health performance and antibiotic use in semi-intensive broiler farms in Uganda. One Health 20: 101039. Available from: https://www.sciencedirect.com/science/article/pii/S2352771425000758?via%3Dihub#s0055
- Kemunto, N.P., Muloi, D.M., Ibayi, E.L., Njaramba, J.K., Hoffmann, V., Murphy, M., Nielsen, S.S. and Moodley, A. 2025. Analysis of antibiotic use, biosecurity and mortality in semi-intensive broiler farms in Kenya. BMC Veterinary Research 21: 541.Available from: https://link.springer.com/article/10.1186/s12917-025-04948-w#Sec3
- Otieno WA, Nyokabi NS, Wanza P, et al. Adoption of farm biosecurity practices among smallholder poultry farmers in Kenya: an application of latent class analysis with a multinomial logistic regression. Prev Vet Med. 2023;218:105974. doi:10.1016/j.prevetmed.2023.105974.
- Venkataramanan V, Crocker J, Karon A, Bartram J. Community-led total sanitation: awareness and its impact on sanitation practices. Int J Community Med Public Health. 2018;5(10):4268-4273. doi:10.18203/2394-6040.ijcmph20183856.
- World Health Organization. Diarrhoeal disease [Internet]. Geneva: WHO; 2024 [cited 2026 Mar 6]. Available from: https://www.who.int/news-room/fact-sheets/detail/diarrhoeal-disease.
- Clemmons EA, Alfson KJ, Dutton CJ. Transboundary animal diseases: an overview of 17 diseases with potential for global spread and serious consequences. Animals (Basel). 2021;11(7):2039. doi:10.3390/ani11072039
- Centre for Affordable Water and Sanitation Technology. Tippy tap: a simple handwashing station for areas with limited water [Internet]. Calgary: CAWST; 2020 [cited 2026 Mar 6]. Available from: https://www.cawst.org/resources/tippy-tap.
- Pickering AJ, Davis J, Blum AG, et al. Access to waterless hand sanitizer improves student hand hygiene behavior in primary schools in Nairobi, Kenya. Am J Trop Med Hyg. 2013;89(3):411-418. doi:10.4269/ajtmh.13-0008.
- Practical Action. Safe pair of hands project report. Rugby: Practical Action; 2021.
- Okello E, Kapisi J, Fleming FM, et al. Factors perceived to facilitate or hinder handwashing among primary students: a qualitative assessment of the Mikono Safi intervention schools in north-western Tanzania. BMJ Open. 2019;9(11):e030947. doi:10.1136/bmjopen-2019-030947.
- Ministry of Health, Kenya. Kenya antimicrobial resistance report 2025. Nairobi: Ministry of Health; 2025.
You may also like
When prevention fails: prescription is inevitable Test article
The first domino in Antimicrobial Stewardship (AMS) has already tipped and initiated a cascade of ev...
Read More

No comments yet. Be the first to comment!